 
Consider theLewis structure for glycine, the simplest amino acid:Ī). Which of the molecules listed in Problem #14.1 above are polar and which are nonpolar? Use simple structure and bonding models to account for each of the following.Ī). Draw the Lewis structure of each of the elemental forms of oxygen, ozone, sulfur, carbon (simple sketch of graphite or diamond) and silicon.ĪLL work must be shown in all problems for full credit. Describe some of the physical properties (phase, color, melting point, boiling point) of oxygen, ozone, sulfur, carbon and silicon.ġ8. Explaining the meaning of the term delocalized as it applies to binding electrons and how the term is used to explain the structure of benzene, C 6H 6.ġ7. Which hybrid orbitals are used to describe multiple bonding between carbon and oxygen in the following molecules?ġ6. Complete the following table by filling in the missing information.ġ5. Given four atomic orbitals, an s orbital and three p orbitals and the resulting set of four sp 3 hybrid orbitals, explain the difference in shape, orientation, size andġ4. Clarify the distinction between the overlaps by drawing a picture of two 'p' orbitals forming a sigma ( s) bond and two 'p' orbitals forming a pi ( p) bond.ġ3. Explain the difference between the model for a sigma ( s) bond and for a pi ( p) bond. Define the term overlap as it relates to interaction of atomic orbitals on atoms.ġ2. Based on a Lewis structure, explain how using the terminal atoms and lone pairs of electrons on the central atom one can distinguish between polar molecules and a nonpolar molecules.ġ1a. How does predicting molecular geometries differ for molecules with multiple 'central atoms' compared to molecules with a single central atom?ĩa.  
In which molecular geometry is it necessary to differentiate between axial and equatorial atoms?Ĩ. (Note: A is the symbol for the central atom, B is the symbol for a bonding group of electrons and N is the symbol for a nonbonding pair of electrons.)ħ. How does the change in the number of bonding electrons and nonbonding electrons affect the molecular geometry for the series AB 4, AB 3N, AB 2N 2. Note: Be sure to include all possible combinations of bonding and nonbonding electrons on the central atom.)Ħ. List the steps used to predict the molecular geometry using VSEPR.ĥ. Differentiate between electron-pair geometry and molecular geometry.Ĥ. Draw the Lewis electron dot structure for CCl 4 and identify all the bonding pairs and nonbonding pairs of electrons.ģ. In your sketch label bond angels and bond lengths.Ģa. All bond angles are equal and all bond lengths are equal. Sketch a five-atom molecule that has a central atom to which the remaining four atoms are independently attached. 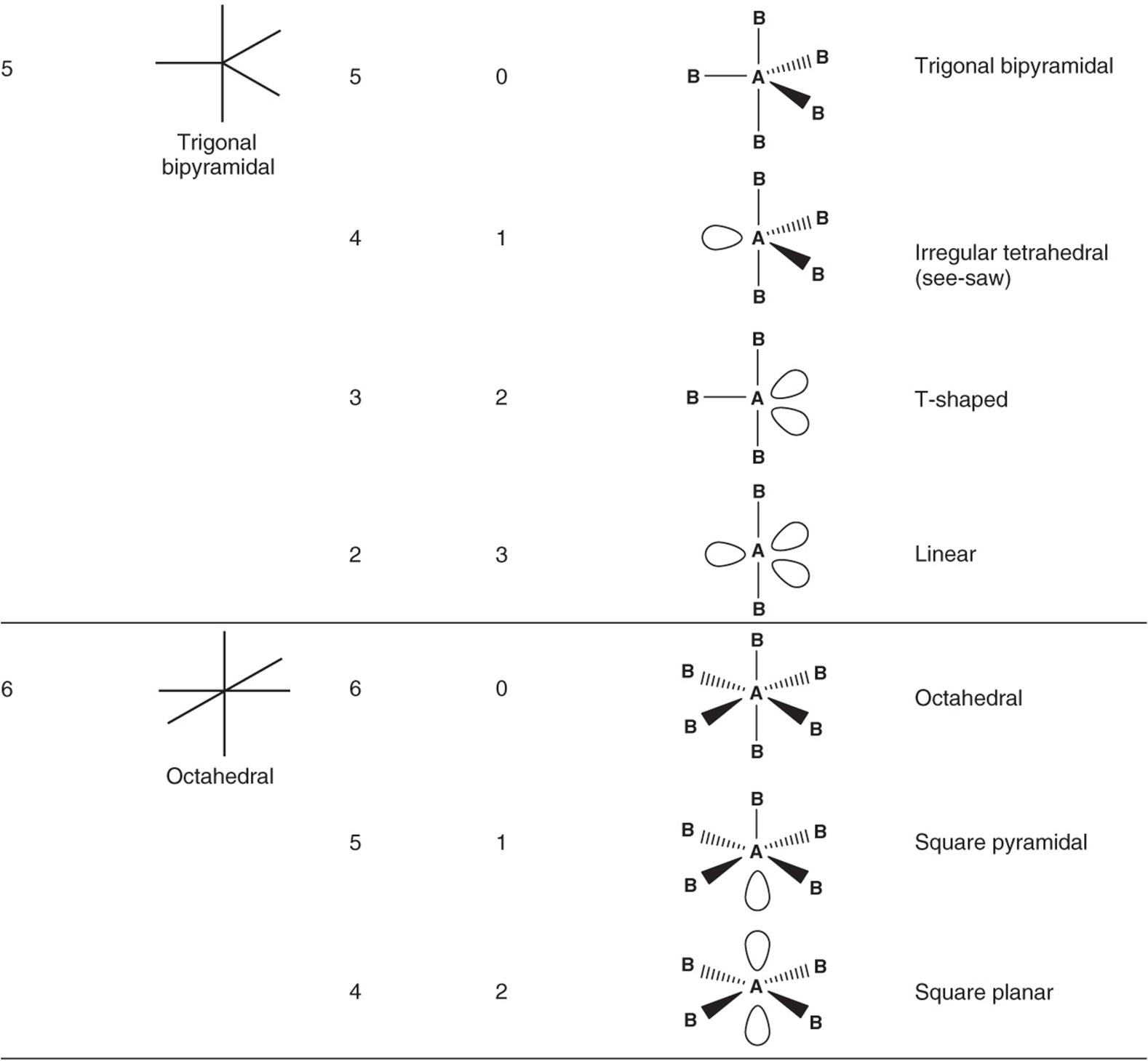
draw and identify the molecular geometry of the elemental form of oxygen, sulfur, carbon and silicon.ġa.list characteristic physical properties of the elemental form of oxygen, sulfur, carbon and silicon.explain the meaning of the concept of delocalized bonding electrons.identify the atomic orbitals used to bind atoms in covalent molecules containing multiple bonds.show how the geometry of a covalent molecule to a particular set of hybrid orbitals on a central atom.describe how a set of hybrid orbitals differ from a collection of atomic orbitals.distinguish between sigma and a pi chemical bonds.define the term overlap and diagram the orbital overlap of two s orbitals, of an s and a p orbital, and of two p orbitals.define the term dipole moment and predict its presence or absence in a covalent molecule.define the term polar and nonpolar as it is used in covalent molecules.identify the molecular geometry about any non-terminal atom in any molecule lacking a single central atom.identify axial and equatorial atoms in simple covalent molecules.describe the effect of nonbonding electrons on bond angle in a given series of covalent molecules.describe molecular geometries that result from bond angles in a given covalent molecule.apply the molecular geometry using valence-shell electron pair repulsion (VSEPR) model to predict molecular geometry.differentiate between the electron-pair geometry and the molecular geometry for any simple covalent molecule.distinguish between bonding pairs and nonbonding pairs of electrons in the molecules of covalent compound.define the terms bond angle and bond length in the molecules of covalent compounds.Following your study of this chapter, you should be able to 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
